Study Reveals Protective Effects of Targeting Intestinal MyD88 on Intestinal Oxidative Injury-induced Weight Loss
Gut health plays a vital role in the overall health and disease control of human and animals. Intestinal oxidative stress is a critical player in the induction and progression of cachexia which is conventionally diagnosed and classified by weight loss. Therefore, reduction of intestinal oxidative injury is a common and highly effective strategy for the maintenance of human and animal health.
Myeloid differentiation factor (MyD88) plays a pivotal role in directing the oxidative and inflammatory response to various stimuli. Autophagy can be regarded as a secondary defense to oxidative stress, by its ability to remove damaged substrates which may accumulate during this process.
Recently, a research team from Institute of Subtropical Agriculture, Chinese Academy of Sciences and Hunan Agricultural University revealed that intestinal MyD88 deficiency ameliorated intestinal oxidative injury. The main findings of this study include: 1) Intestinal MyD88 deficiency ameliorated intestinal mitochondrial dysfunction and DNA damage during PQ exposure; 2) There was a marked decrease in ROS in MyD88-deficient cells during PQ exposure, which were consistent with high activity of antioxidative enzymes; 3) Intestinal MyD88 deficiency effectively ameliorated weight loss caused by intestinal oxidative injury in an autophagy-dependent manner.
This work was supported by the National Natural Science Foundation of China (32072745 and 32130099) and Innovation Province Project (2019RS3021). The paper entitled "MyD88 deficiency ameliorates weight loss caused by intestinal oxidative injury in an autophagy-dependent mechanism" has been published in Journal of Cachexia, Sarcopenia and Muscle.
More details could be found at https://onlinelibrary.wiley.com/doi/10.1002/jcsm.12858
Contact: Yulong Tang
E-mail: tangyulong@isa.ac.cn
Institute of Subtropical Agriculture, Chinese Academy of Sciences
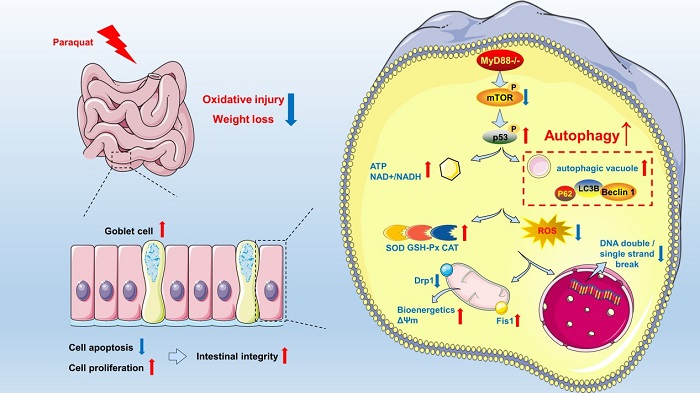
Figure. Proposed mechanisms underlying MyD88 deficiency ameliorates weight loss caused by ROS-mediated intestinal mitochondrial dysfunction and DNA damage through promoting autophagy.(Image by Ming Qi)
Download attachments: