Researchers Reveal Iron-Carbon Coupling Mechanisms for Enhancing Carbon Sequestration in Paddy Soils
A research team led by Prof. Wu Jinshui from the Institute of Subtropical Agriculture, Chinese Academy of Sciences has decoded the intricate iron-carbon interactions governing soil organic carbon (SOC) stability, unveiling novel strategies to amplify agricultural carbon sequestration.
Rice paddies, critical for global food security, also serve as vital carbon sinks with untapped potential to mitigate climate change. Iron minerals stabilize approximately 33.5% of global SOC through chemical bonding and coprecipitation. However, microbial iron reduction in waterlogged paddies can destabilize this stored carbon, releasing it back into the atmosphere.
In their groundbreaking study, they discovered that rice roots form iron-rich plaques through oxygenation-driven Fe(II) oxidation, which trap organic carbon in non-crystalline iron matrices resistant to microbial breakdown. Field experiments showed that adding Fe(II) boosts plaque formation, sequestering an estimated 130 million metric tons of carbon globally per rice-growing season. This “rust sink” mechanism replenishes SOC pools and highlights a natural pathway to offset agricultural emissions (Fig. 1).
The team investigated the fate of iron-organic carbon complexes in the anaerobic paddy soils, by employing 13C-labeled iron-glucose complexes. They demonstrated that high-crystallinity iron minerals like 6-line ferrihydrite reduce carbon mineralization by 21% compared to low-crystallinity forms. Such minerals also suppress SOC decomposition by limiting microbial access and inducing phosphorus scarcity, enhancing net carbon storage by 51% (Fig. 2).
The stability of iron-carbon complexes was found to depend on mineral type and binding modes. Goethite, a high-crystallinity iron oxide, outperformed other minerals when coprecipitated with organic carbon, slashing CO₂ and CH₄ emissions by 17%–41% and 21%–61%, respectively (Fig. 3). This binding mode reduced the global warming potential by up to 71%. The findings emphasize that iron oxides act as dual regulators-serving as electron acceptors in microbial processes while immobilizing nutrients to suppress decomposition.
Collectively, these findings systematically elucidate the protective mechanisms of iron oxides on rhizodeposited carbon in paddy soils, and clarify the synergistic regulatory mechanisms of iron oxide crystallinity, organic carbon binding modes, and carbon loading capacity on SOC accumulation. “From an agricultural management perspective, promoting the coprecipitation of high-crystallinity iron oxides with organic carbon not only enhances SOC sequestration capacity but also significantly mitigates the contribution of paddy ecosystems to global warming by reducing greenhouse gas emissions such as CH,” said by Dr. Duan Xun, the first author.
Their findings were published in Global Change Biology (2022), Soil Biology and Biochemistry (2023, 2025), and Science of the Total Environment (2024).
Contact: Wu Jinshui
Email: jswu@isa.ac.cn
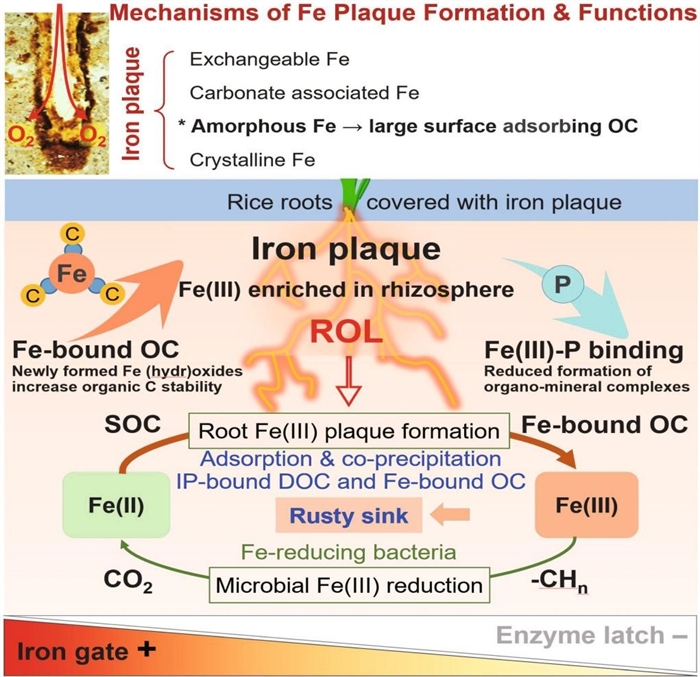
Figure 1 Biotic and abiotic mechanisms of carbon trapping by root iron plaque (Imaged by Wei Liang)
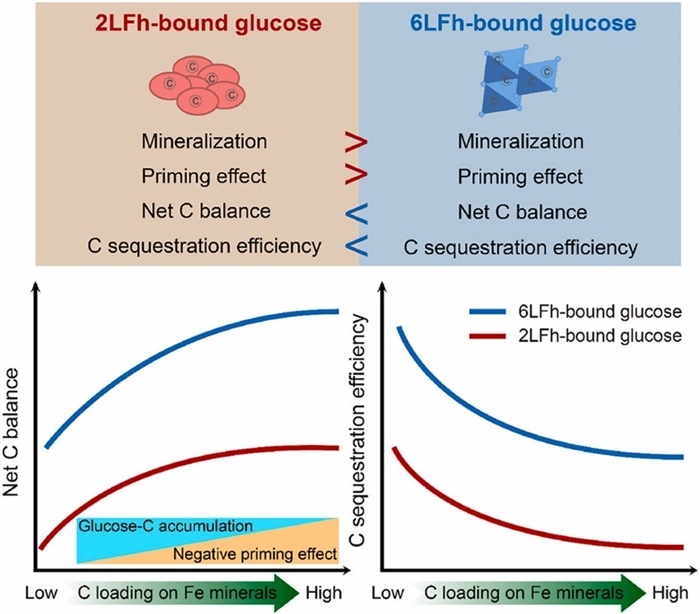
Figure 2 Variation patterns of carbon sequestration efficiency in iron-carbon complexes with respect to iron mineral crystallinity and carbon loading capacity (Imaged by Duan Xun)
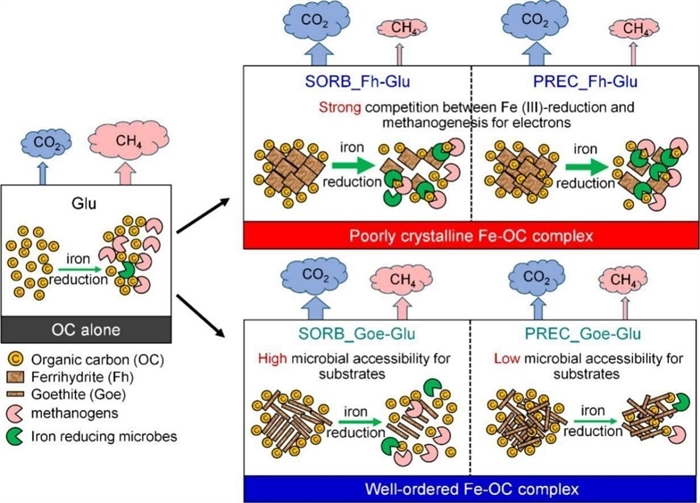
Figure 3 Impact of iron-carbon complex binding forms on the release and mineralization of associated carbon under anaerobic conditions (Imaged by Gao Wei)
Download attachments: